


IN THE HIGH COURT OF JUDICATURE AT ALLAHABAD
HARVIR SINGH
Marion Biotech Pvt. Ltd. – Appellant
Versus
Union of India – Respondent
JUDGMENT :
HARVIR SINGH, J.
1. Heard Sri Niraj Kumar Singh, holding brief of Sri Saroj Kumar Yadav, learned counsel for the revisionists, and Sri R.P.S. Chauhan, learned counsel for the opposite party-Union of India, and perused the record.
2. This Criminal Revision is directed against the impugned cognizance and summoning order dated 19.01.2024 passed by the learned Chief Judicial Magistrate, Gautam Budh Nagar in Complaint Case No. 2462 of 2024 (Union of India vs. Ms Marion Biotech Pvt. Ltd. & Others), under Sections 18(a)(i), 16, 17-A, 17-B, 18-A, 18-B and punishable under Sections 27(a), 27(b)(i), 27(b)(ii), 27(c), 27(d), 28, 28-A, and 28-B of the Drugs and Cosmetics Act, 1940, Police Station Phase III, Noida, District Gautam Buddh Nagar.
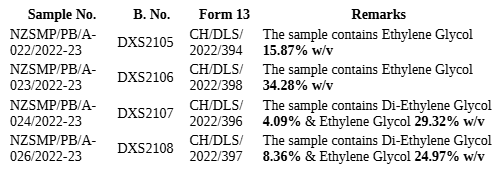
3. The brief facts of the case, are that the revisionists, being directors and officials of M/s. Marion Biotech Pvt. Ltd., (hereinafter referred as Company) they were summoned, pursuant to a complaint filed by the Drugs Inspector, alleging various violations including manufacture/sale of drugs declared "not of standard quality" and further invoking sections related to adulterated and spurious drugs, procedural non-compliance, and liability of compa

Directors are liable under the Drugs and Cosmetics Act for the conduct of the company's business, and summoning orders require only a prima facie case without extensive procedural scrutiny.

Non-compliance with statutory provisions and resignation of the accused from the company absolved him of liability, leading to the quashing of the proceedings.

Directors of a company not involved in drug manufacturing cannot be held liable under the Drugs and Cosmetics Act absent specific evidence of their responsibility for the conduct of business.

Compliance with statutory provisions is essential for the validity of prosecution actions; failure to adhere to such requirements can lead to acquittal.

Non-compliance with inspection procedures under the Drugs and Cosmetics Act, 1940 is not fatal for prosecution in cases involving allegations of spurious drugs.

The main legal point established in the judgment is that the prosecution for manufacturing drugs not of standard quality may not be launched if the product is free from any foreign matter and the def....

Prosecution quashed for delay in sample testing beyond 60 days without extension under Rule 45 and failure to send sample to manufacturer under Section 23(4)(iii), depriving re-analysis right post sh....

Conviction under drug regulation provisions requires proving non-compliance, highlighting the importance of disclosing the manufacturer's details for legal protection.


The court's decision emphasized the importance of considering the actual quality of the product, the timing of the launch of prosecution, and the availability of administrative measures for minor def....


Login now and unlock free premium legal research
Login to SupremeToday AI and access free legal analysis, AI highlights, and smart tools.
Login
now!
India’s Legal research and Law Firm App, Download now!

Copyright © 2023 Vikas Info Solution Pvt Ltd. All Rights Reserved.



